SFEBES2012 Poster Presentations Reproduction (23 abstracts)
Periodic exposure to exogenous testosterone is required to maintain long-term integrity of the GnRH pulse-generator in a male with atypical Kallmann syndrome
Anjali Santhakumar 1 & Richard Quinton 1,
1Endocrine Unit ,Royal Victoria Hospital, Newcastle Hospitals NHS, Newcastle upon tyne, United Kingdom; 2Institute of Genetic Medicine, University of Newcastle upon Tyne, Newcastle upon tyne, United Kingdom.
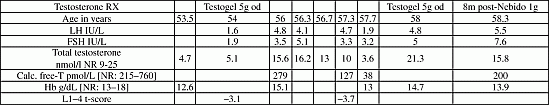
Kallmann syndrome is a treatable form of male infertility caused by a congenital defect in the secretion or action of gonadotropin releasing hormone (GnRH). It can permanently remit in around 10% of male cases. We report a patient with atypical Kallman’s syndrome who requires intermittent testosterone replacement to sustain activity of his gonadotrophic axis A 55 year old male presented with erectile dysfunction and decreased libido. He reported normal puberty, with no history of cryptorchidism. Examination revealed marked segmental disproportion with anosmia -but no clefting or synkinesis. Testis volume was 15 mL. Biochemistry confirmed isolated hypogonadotrophic hypogonadism, MRI demonstrated hypoplastic olfactory sulci. DEXA scan evidenced spinal osteopaenia. Research-based screening was negative for coding sequence mutations of: FGF8, FGFR1, GNRH1, GNRHR, KAL1, KISS1R, KISS1, NELF, PROK2, PROKR2, TAC3 and TACR3. A clinical diagnosis of atypical Kallmann’s syndrome was made and he was started on Testogel 10 g daily with good clinical improvement. Reassessment off therapy after 2 years evidenced recovery of gonadotrophic function (Table 1). However, over the subsequent 2 years; off therapy his gonadotrophic axis progressively waned accompanied by decrease in bone density. He was restarted on Testosterone undecanoate 1 g IM-(Nebido). Initially planned frequency of 3–4 months was postponed due to persistently normal serial testosterone levels. We have since determined that he requires Nebido 1g IM every 8 months in order to maintain optimal serum T and LH pulsatility.
Conclusion: This patient seems to have a “vulnerable” gonadotrophic axis that is prone to shutting-down. However, intermittent exposure to exogenous Testosterone suffices to maintain long-term integrity of this axis, presumably through androgen-related upregulation of genes and/or systems involved in central reproductive control.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.