SFEBES2012 Oral Communications Young Endocrinologists prize session (8 abstracts)
Functional characterisation and translational clinical applications of kisspeptin-10
Jyothis George 1 , Richard Quinton 4 , Jacques Young 5 , Johannes Veldhuis 3 , Robert Millar 2 & Richard Anderson 1
1MRC Centre for Reproductive Health, University of Edinburgh, Edinburgh, United Kingdom; 2Centre for Integrative Physiology, University of Edinburgh, Edinburgh, United Kingdom; 3Endocrine Research Unit, Mayo Clinic, Rochester, MN; 4Institute of Genetic Medicine, Newcastle University, Newcastle, United Kingdom; 5Service d’Endocrinologie et des Maladies de la Reproduction, Hôpital de Bicêtre, Paris, France.
Hypothesis: Exogenous kisspeptin-10 (Kp10) enhances pulsatile LH. Research strategy: A first-in-man dosing study of kp10 was first performed. Healthy volunteers and hypogonadal patients were subsequently infused with kp10. Dose titration study Rapid increases in LH with clear dose-dependency (P<0.0001) were observed in healthy men (n=6), with the 0.3 and 1 µg/kg doses being maximally stimulatory (P<0.01). Effect of sex-steroid milieu: LH responses to iv Kp10 (0.3 µg/kg,) in the follicular phase (n=10) was compared with that in postmenopausal women (n=6) and in women taking combined contraceptives (n=8). LH increased significantly and comparably following Kp10 in the follicular phase (6.3±1.2 to 9.4±1.3 IU/L P=0.006), postmenopausal (35.3±2.8 to 44.7±3.4 IU/L P=0.005) and COCP groups (2.2±0.9 to 3.7±1.4 IU/L P<0.001). Pulse-frequency study: LH pulses increased in healthy men from 0.7±0.1 to 1.0±0.2 pulses/hr (P = 0.01) during a 12 hr 4 µg/kg per hr infusion, with LH increasing from 5.2±0.8 to 14.1±1.7 IU/L (P<0.01). High dose infusion study: Four healthy men were admitted for 34 hrs and an infusion of Kp10 (4 µg/kg per hr) maintained for 22.5 hrs. LH increased from 5.5±0.8 to 20.9±4.9 IU/L (P <0.05) and testosterone from 16.6±2.4 to 24.0±2.5 nmol/L (P <0.001). Studies in hypogonadal patients: An infusion of kp10 (4 mcg/kg per hr) increased LH (3.9±0.1 to 20.7±1.1 IU/L (P=0.03) and testosterone (8.5±1.0 to 11.4±0.9 nmol/L, P=0.002) in T2DM. LH pulse-frequency at baseline was lower in T2DM (0.6±0.1 vs. 0.8±0.1 pulses/hr, P=0.03) and increased to 0.9±0 pulses/hr (P=0.05. Mean LH and LH pulse-frequency increased with kisspeptin-10 (P<0.05) in patients with TAC3 or TACR3 mutations. However, individuals with Kallmann syndrome did not respond.
Summary of LH Responses to kisspeptin-10 and GnRH Kisspeptin-10 (0.3 mcg/kg) was administered as bolus with 15-min blood sampling for 3 hours before and after.

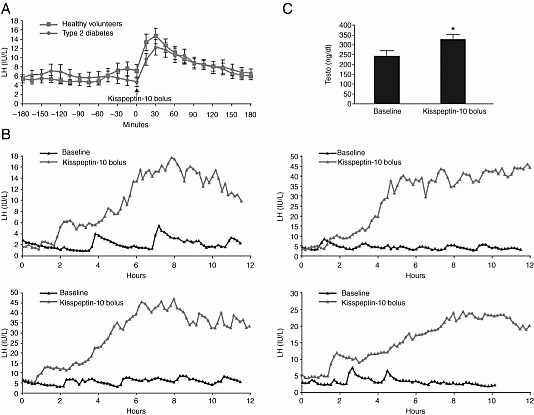
Conclusions: Kisspeptin-10 increases LH pulse frequency. Proof-of-concept studies demonstrate potential clinical applications for this novel peptide.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.

 }
}



