SFEBES2012 Poster Presentations Pituitary (43 abstracts)
Ipilimumab induced hypophysitis - an evolving clinical entity
Agata Juszczak , Niki Karavitaki & Ashley Grossman
Oxford Centre for Diabetes, Endocrinology and Metabolism, Oxford University Hospitals, Oxford, United Kingdom.
Introduction: Ipilimumab (Yervoy, BMS) is a human monoclonal antibody increasingly used in oncology. It targets cytotoxic T lymphocytes antigen 4 (CTLA-4) promoting their proliferation and augmenting an anti-tumour response. It is licensed for the treatment of unresectable or metastatic melanoma and multiple clinical trials using this medication for other malignancies are ongoing. Ipilimumab related adverse reactions are mainly autoimmune and occur in 61% of patients, of which 7.6% involve endocrinopathies. Case report A 54 year-old man presented to the oncology clinic with headaches and severe lethargy following third course of ipilimumab for recurrent metastatic melanoma. He had been previously treated with local wide excision and chemotherapy, which failed to control the disease. He did not have other co-morbidities. As metastatic brain disease was suspected, an MRI scan was arranged, which showed a pituitary oedema consistent with hypophysitis. Investigations demonstrated anterior pan-hypopituitarism (morning serum cortisol 10 nmol/l, testosterone <4 nmol/l with low normal LH and FSH, TSH < 0.01 mU/L, fT4 9.4 pmol/L). There was no diabetes insipidus. He was started on dexamethasone, L-thyroxine and testosterone replacement with rapid resolution of his symptoms. The dose of dexamethasone was gradually decreased and replaced with hydrocortisone 10/5/5 mg. A repeat MRI scan is planned for three months’ time.
Discussion: In a phase III clinical trial of ipilimumab, known as MDX010–20, hypophysitis occurred in 1.5% of patients and hypopituitarism in 2.3%. Other endocrinopathies included hypo- and hyperthyroidism secondary to thyroiditis, primary adrenal insufficiency and isolated hypogonadism. In the literature single case reports have described ipilimumab-related SIADH, hypophysitis with diabetes insipidus and a Graves’ ophthalmopathy-like syndrome.
Conclusions: Endocrinologists need to be rapidly aware of new immuno-modulating agents used in cancer treatment as they cause a variety of autoimmune complications which could be life threatening if unrecognised.
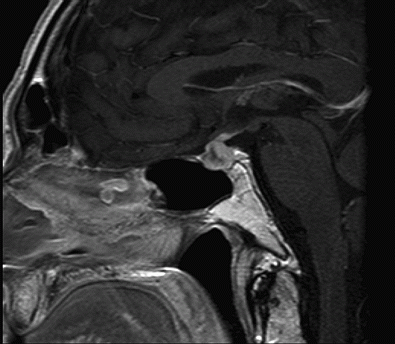
MRI scan showing pituitary oedema consistent with hypophysitis.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.
 }
}



