SFEBES2012 Poster Presentations Reproduction (23 abstracts)
An audit on investigative workup of patients with suspected secondary hypogonadism
Logan Manikam , Mohammed Kazi , Ananth Nayak & Harit Buch
Wolverhampton Diabetes Centre, Royal Wolverhampton Hospitals NHS Trust, Wolverhampton, United Kingdom.
Background: Testosterone deficiency (TD) affects 5–6% of men aged 30–79 years and it can be a presenting feature of a serious underlying condition like a pituitary tumour. Clinical practice guidelines have recommended thorough investigative workup of such patients to delineate the underlying pathology in patients with secondary TD. We have undertaken a retrospective study to assess the need for expensive investigations in patients with relatively mild TD suspected to have a central cause.
Methods: Electronic data and notes of 114 patients who had serum testosterone estimation between January and December 2010 were reviewed. Patients <25 years old and patients with established hypothalamo-pituitary pathology or primary TD were excluded. 30 patients were suspected to have secondary TD (normal or low gonadotrophins) and underwent further complete pituitary function tests. Patients were divided into 2 sub-groups i.e. ≥5 nmol/l (Group-A) and <5 nmol/l (Group-B) (reference range: 7.6–19 nmol/l) and both groups were compared for the outcomes of pituitary function tests and imaging studies.
Results: Number of patients in Group-A and Group-B were 22 and 8 and mean (median) testosterone was 7.9(8.1) and 2.3(2.3) nmol/l respectively. Group-B had a significantly higher proportion of patients with abnormal pituitary function tests in comparison to Group-A (62% v 14% respectively, P=0.02). In all, 14 patients underwent pituitary magnetic resonance imaging (MRI) scanning. Only 1/6 patients in Group-A in comparison to 4/8 patients in group-B had an abnormal scan. None of the patients with normal pituitary function tests in either of the two groups had an abnormal MRI scan.
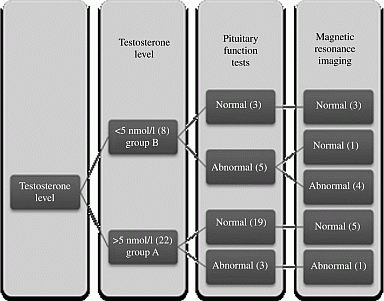
Patients with low testosterone investigation flowsheet
Conclusion: Clinical guidelines currently recommend an exhaustive investigative work-up in patients with low testosterone suspected to have secondary TD. Our results suggest that a more selective approach can be considered in patients with milder testosterone deficiency.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.
 }
}



