ICEECE2012 Oral Communications Adrenal Clinical (6 abstracts)
Differential diagnosis of primary aldosteronism by peripheral plasma levels of 18-oxo-cortisol: a noninvasive method with a high specificity
F. Satoh , R. Morimoto , M. Kudo , Y. Iwakura , Y. Ono , K. Takase , Y. Nakamura , H. Sasano & S. Ito
Tohoku University Hospital, Sendai, Japan.
18-Oxocortisol (18-oxoF) is a derivative of cortisol (F) that is produced by aldosterone synthase (CYP11B2), and its clinical usefulness has not been clarified. We prospectively measured 18-oxoF, using highly sensitive liquid chromatography-tandem mass spectrometry (LC/MS/MS), in peripheral plasma of patients with primary aldosteronism (PA) who underwent adrenal venous sampling (AVS) to differentiate aldosterone producing adenoma (APA) from bilateral hyperplasia. The study was performed in 116 patients with surgically-proven APA and 121 patients with bilateral hyperplasia diagnosed by AVS which was successfully performed. The levels of 18-oxoF were significantly higher in the peripheral plasma from the patients with APA than that from the patients with bilateral hyperplasia (bilateral hyperaldosteronism (BHA) diagnosed by AVS and surgically-proven idiopathic hyperaldosteronism (IHA) were shown in Fig. 1). The cutoff value (4.7 ng/dl) of 18-oxoF levels was calculated by ROC analysis with sensitivity of 0.64 and specificity of 0.97. In the cases with CT-positive unilateral adrenal masses, the cutoff value (4.6 ng/dl) of 18-oxoF levels was calculated by ROC analysis with sensitivity of 0.73 and specificity of 0.97. In these cases, the 18-oxoF levels in 60 cases (80%) with APA were higher than 4.6 ng/dl, and while, those in 29 cases (97%) with bilateral hyperplasia and those in eight cases (89%) with contralateral CT-negative micro-APA were lower than 4.6 ng/dl. Thus, according to peripheral blood levels of 18-oxo-cortisol, 80% of APA patients with unilateral masses might be able to undergo ipsilateral adrenalectomy without AVS.18-oxoF levels can be a clinically useful biomarker for differentiating APA from bilateral hyperplasia.
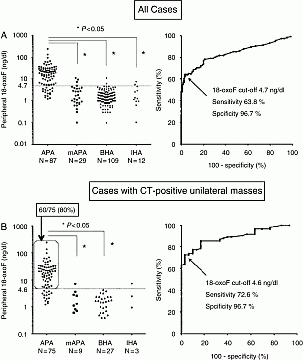
Figure 1 ROC of 18oxoF plasma levels for the identification of APA versus bilateral hyperplasia in all cases (A) and cases with CT-positive unilateral adrenal masses (B): APA; cases with surgically-proven ipsilateral aldosterone producing CT-positive adenoma (A, B): mAPA (A); cases with CT-negative aldosterone producing adenoma: mAPA (B); cases with CT-positive non-functioning adenoma and contralateral CT-negative aldosterone producing adenoma: BHA; bilateral hyperaldosteronism diagnosed by AVS: IHA; surgically-proven idiopathic hyperaldosteronism.
Declaration of interest: The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research project.
Funding: This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
 }
}



