SFEBES2012 Poster Presentations Obesity, diabetes, metabolism and cardiovascular (73 abstracts)
Exposure to elevated glucose prior to differentiation re-programs the cellular secretory pathway in fetal hepatocytes
David Gardner 1 , Catherine Wells 2 & Simon Welham 2
1School of Veterinary Medicine, University of Nottingham, Loughborough, United Kingdom; 2Division of Nutritional Sciences, University of Nottingham, Loughborough, United Kingdom.
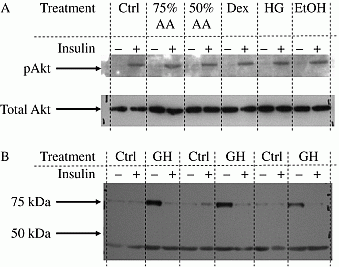
Various programming stimuli have been postulated to permanently influence fetal development with the common result that offspring exhibit differing degrees of insulin resistance. We set out to establish whether protein restriction, high glucose, glucocorticoid or alcohol exposure might exert their effects directly using a derived population of fetal hepatocytes. Cells were isolated from fetal mouse livers (n=10) at embryonic day 12. Fetal livers were rinsed in phosphate buffered saline, mechanically disrupted into a single cell suspension and plated onto plastic tissue culture flasks. Cells were grown prior to differentiation in a variety of media which varied in either essential amino acid content (75%AA, 50%AA), glucose content (HG - 20mM glucose) or included either dexamethasone (Dex) or ethanol (EtOH). All cells were differentiated with dexamethasone and retinoic acid after treatment. Insulin sensitivity in differentiated cells as measured by insulin induced Akt phosphorylation showed no influence of pre-differentiation programming (Figure 1A). Measurement of α-fetoprotein to determine the degree to which programming stimuli may have altered the hepatic phenotype revealed two isoforms (~75kDa & ~42KDa). The 75KDa isoform showed insulin stimulated depletion exclusively in high glucose treated cells. Casein zymography did not vary with pre-differentiation treatment. Blockade of protease activity did not alter zymography results, but enhanced the α-fetoprotein response to insulin (Figure 1B), indicating that α-fetoprotein depletion was not mediated by proteolytic activity. These results suggest that, whilst all of the “programming” stimuli failed to directly impact upon fetal hepatic insulin sensitivity, exposure to elevated glucose prior to differentiation appeared to establish a novel mechanism of insulin regulated control of the secretory pathway. The isoform specificity of this may indicate that a restricted population of proteins may be subject to such programmed regulation.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.
 }
}



