SFEBES2012 Poster Presentations Clinical biochemistry (15 abstracts)
An unusual case of hypocalcaemia
Ye Kyaw 1 , Jennifer Tringham 1 , Tim Wang 2 & Emma Bingham 1
1Endocrinology, Frimley Park Hospital, Camberley, United Kingdom; 2Biochemistry, Frimley Park Hospital, Camberley, United Kingdom.
An 87 year old gentleman with type 2 diabetes mellitus was started on oral sodium fusidate for infected left middle toe ulcer. His blood test revealed Sodium 140 mmol/L, potassium 5.2 mmol/L, urea 25.3 mmol/L, creatinine 287 µmol/L, eGFR-19 ml/minute, albumin 29 g/L, adjusted calcium 2.17 mmol/L, and phosphate 0.95 mmol/L. Two days later he was admitted to hospital. Blood tests on admission showed severe hypocalcaemia with renal failure. (Sodium 146 mmol/LL, potassium 4.9 mmol/L, urea 26.7 mmol/L, and creatinine 236 µmol/L, eGFR 23 ml/minute, adjusted calcium 1.6 mmol/L and phosphate 1.34 mmol/L). He did not have any signs and symptoms of hypocalcaemia. Despite treatment with intravenous calcium and oral vitamin D tablets, his calcium level did not normalize. His PTH was 37. On day 3, oral sodium fusidate was stopped and serum calcium subsequently normalized. Discussion Two cases of hypocalcaemia related to fusidic acid were reported by Biswas et al. [1] In both cases, hypocalcaemia was associated with renal failure and hepatic impairment. They postulated that hypocalcaemia secondary to fusidic acid was likely to be due to impaired vitamin D synthesis in liver and renal failure. A case of hypocalcaemia was also reported after high dose intravenous fusidic acid and hypocalcaemia was thought to be due to high phosphate contents in the intravenous antibiotics. [2] In our case, liver enzymes were normal. It seems that renal failure is more likely precipitant for hypocalcaemia in fusidic acid antibiotic therapy. From a clinician's perspective, it is imperative to monitor serum calcium level with fusidic acid therapy particularly in the presence of renal failure. References: [1] Biswas M, Owen K, Jones MK. Hypocalcaemia during fusidic acid therapy. Journal of the Royal Society of Medicine. 2002 Feb;95(2):91–93. [2] Cowan JC HS. Profound hypocalcaemia after high doses of intravenous fusidic acid. British Medical Journal. 1984;288:684.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.
Blood results
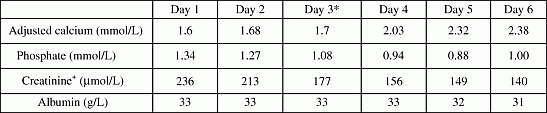
*day 3 = sodium fusidate was stopped. + Renal failure was treated conservatively with intravenous fluid.
 }
}



