SFEBES2012 Poster Presentations Thyroid (52 abstracts)
Predicting risk of relapse following thionamide withdrawal in Graves’ disease
Fraser Gibb , Mark Strachan & Nicola Zammitt
Edinburgh Centre for Endocrinology, Edinburgh, United Kingdom.
Background: Male sex, cigarette smoking, young age, severe thyrotoxicosis, large goitre and marked or persistent elevation of TRABs are consistently reported as predictors of relapse following withdrawal of thionamides in Graves’ disease. Early recurrence rates are commonly quoted at 50% but the definition of early recurrence varies widely across the literature. AIM We sought to determine the best clinical predictors of recurrent thyrotoxicosis and whether these would permit stratification of risk of recurrence.
Methods: Retrospective review of 201 patients who completed a course of carbimazole (or PTU) and had at least one year of follow-up TFTs. Sex, age, free T4, total T3, initial TRAB, Anti-TPO antibody, time to normalisation of TSH and TRAB levels at cessation of therapy were assessed and thyroid status one year after treatment cessation was recorded (relapse or remission). Logistic regression analysis identified significant predictors of relapse and a model for predicting early recurrence risk was constructed.
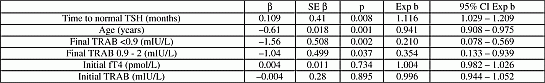
Logistic regression assessing risk of recurrence at one year (Nagelkerke R2 0.208). TRAB categories <0.9 and 0.9–2 are referenced against TRABS >O2. Shorter time to normalisation of TSH, lower TRABs and older age were associated with higher remission rates.
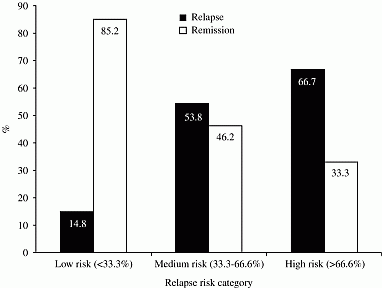
Results: Relapse of thyrotoxicosis occurred in 27% of patients at one year. Age at presentation, time to normalisation of TSH and TRAB level at cessation of therapy were the best predictors of relapse at one year (table) and were incorporated into a relapse risk calculator. Patients categorised as low risk were very unlikely to relapse within one year (figure). Discussion This cohort is likely to be representative of the types of patients in whom withdrawal of thionamide is considered (i.e. does not include patients with gross persistent elevation of TRABs). We have shown that a set of simple clinical parameters can help categorise the risk of recurrence, which may be of use in individualising follow-up and in better informing patients.
Declaration of interest: There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding: No specific grant from any funding agency in the public, commercial or not-for-profit sector.

 }
}



