ICEECE2012 Poster Presentations Obesity (114 abstracts)
Aldosterone increases oxidative stress in differentiated, but not in undifferentiated 3T3-L1 adipocytes via activation of mineralocorticoid receptors
R. Limor , M. Uda , E. Knoll & N. Stern
Tel Aviv Sourasky Medical Center and Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel.
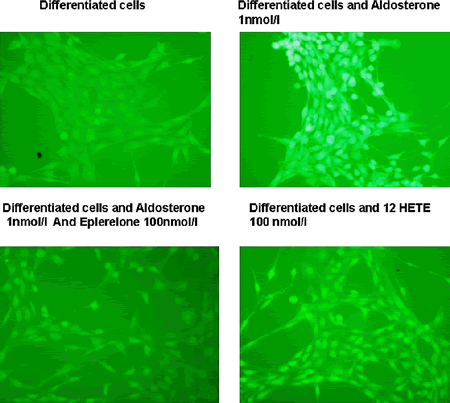
In the cardiovascular system, aldosterone promotes inflammation, tissue remodeling and endothelial dysfunction. In diet-induced obesity, lipid accumulation involves acceleration of preadipocyte differentiation into mature adipocytes. Mutually exerted control mechanisms may exist between aldosterone secretion and fat: circulating aldosterone is increased in proportion to fat mass in humans and fat tissue expresses mineralocorticoid receptors (MR). Here we tested whether or not aldosterone affects oxidative stress in 3T3-L1 cells, a rodent line of preadipocytes which undergoes differentiation to mature fat cells under the combined influence of serum, dexamethasone and insulin. First, both undifferentiated and differentiated 3T3-L1 cells expressed MR as quantified by Real Time PCR. Second, aldosterone (at a physiological concentration-1 nmol/l) had no effect on cell differentiation as assessed by the expression of fat cell differentiation markers, including AP2 and PPARγ. Third, aldosterone (1 nmol/l) induced a ~3.5 increase in the formation of intracellular reactive oxygen species (ROS) as determined by oxidative conversion of cell-permeable chloromethyl-2′,7′-dichlorodihydroflurescein diacetate to fluorescent dichlorofluorescein (DCF) in both differentiating and fully differentiated adipocytes but not in undifferentiated 3T3-L1 preadipocytes. This effect was fully blocked by the specific MR antagonist eplerelone (100 nmol/l). We identified three isoforms of lipoxygenase (LO), dioxygenase enzymes which incorporate molecular oxygen into unsaturated fatty acids such as arachidonic acid and linoleic acid in these cells, platelet 12(S)-LO, leukocyte type (12/15)- and an epidermal LO, of which the expression of the platelet and leukocyte type increased with aldosterone treatment. One LO product, 12 hydroxyeicosatetraenoic acid (12HETE) was able to increase ROS formation in differentiated 3T3-L1 cells (X2-3 folds). These results suggest that aldosterone can increase oxidative stress in differentiated, but not undifferentiated adipocytes, indicating that fat accumulation in mature fat cells predisposes adipose tissue to the pro-oxidative effect of aldosterone, and perhaps serves as a means to increase ROS through oxidation of fatty acids such as 12HETE.
Declaration of interest: The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research project.
Funding: This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
 }
}



