SFEBES2013 Poster Presentations Clinical biochemistry (22 abstracts)
Glycosylated linkers to generate long-acting GH tandems
Ian Wilkinson 1 , Pippa Cawley 1 , Maximilian Bielohuby 2 , Jon Sayers 1 , Peter Artymiuk 1 , Martin Bidlingmaier 2 & Richard Ross 1
1Sheffield University, Sheffield, UK; 2Endocrine Research Unit, Medizinische Klinik und Poliklinik IV, Munich, Germany.
Background: The development of recombinant biologics has had a major impact on many diseases. However, most biologics are rapidly cleared from the body and therefore require frequent injection regimens. There is therefore a need for technologies that allow the half-lives of these molecules to be extended in a predictable manner.
Hypothesis
Increasing numbers of N-linked glycosylation motifs between two GH molecules leads to gradually increased half-life whilst retaining biological activity.
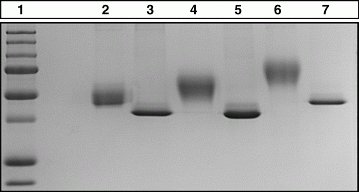
Methods: A number of GH tandem molecules with linkers containing 2–8 NAT glycosylation motifs and their respective controls (in which N is replaced by Q in the sequence motif NAT) were cloned, sequenced and expressed in a CHO cell line. SDS–PAGE was used to verify increases in molecular weight and an in house dual luciferase reporter assay used to test bioactivity. Protein was purified using immobilised metal affinity chromatography (IMAC). Pharmaco kinetics (PK) were assessed using a rat model system.
Results: On increasing numbers of glycosylation motifs a concomitant increase in molecular weight (MW) as observed by SDS–PAGE (see Fig. 1) from 42 to 75 kDa (n=8 glycosylations). All GH tandems were purified using IMAC from suspension-adapted serum free cultures to >95% purity. PK was tested in tandem GH molecules with and without two glycosylation sites. The tandem without glycosylation was cleared rapidly and with an identical profile to monomeric GH whilst the tandem with glycosylation showed a fourfold slower clearance.
Conclusion: It is possible to increase the apparent MW of hormone tandems using glycosylated-linkers whilst maintaining bioactivity and that this also delays clearance of the molecule in rat model system providing a potential technology for generating long acting biologicals.
 }
}



