NuclearReceptors2018 Oral Communications (1) (8 abstracts)
Chemical systems biology analyses reveal dissociated glucocorticoid signaling networks in skeletal muscle
Nelson E Bruno 1 , Sathish Srinivasan 1 , Jerome C Nwachukwu 1 , Zhuang Jin 2 , Jason Nowak 1 , Theodore M Kamenecka 2 & Kendall W Nettles 1
1Department of Structural and Computational Biology, The Scripps Research Institute, Jupiter, Florida 33458, USA; 2Department of Molecular Medicine, The Scripps Research Institute, Jupiter, Florida 33458, USA.
Glucocorticoids (GC) are catabolic in skeletal muscle to provide nutrients during fasting or other stressors, including inhibiting insulin-mediated glucose uptake, protein synthesis, and mitochondrial function, while stimulating proteosomal breakdown of proteins. Published work in cultured myotubes required μM GC dosing to see these effects. We found GCs to show low nM activity in myotubes by using a more physiologically relevant setting, including nutrient deprivation and insulin challenge. In order to understand the molecular basis for these phenotypic activities we generated quantitative, statistically robust bioassays in myoblasts and myotubes and characterized a set of GCs designed to perturb the glucocorticoid receptor with several distinct structural mechanisms. The ligands displayed a full range of variance across the skeletal muscle bioassays, allowing us to identify ligand-specific gene expression patterns that were highly predictive for their effects on insulin-mediated phosphorylation of AKT, glucose disposal, mitochondrial function, and protein balance, including protein synthesis and proteosomal degradation. In vivo validation reveals that our approach, called ligand class analysis, can tie chemical and receptor structure to specific transcriptional signaling outcomes that define glucocorticoid action in skeletal muscle. In doing so we identified a dissociated glucocorticoid with full anti-inflammatory activity that is slightly anabolic for protein balance and mitochondria, and a full antagonist with strong anabolic activity.
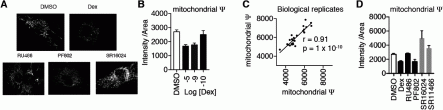
Figure 1 Identification of glucocorticoids with beneficial effects on mitochondria. A) C2C12 myoblasts were starved for 24 hr during treatment with the indicated compounds and analyzed with high content imaging after labeling using mitotracker dye. B) Mitochondrial potential was quantitated as the intensity of mitotracker dye/mitochondrial area. C) Assay reproducibility with 22 novel glucocorticoids + controls. D) Quantitation of A) shows that PF802, a dissociated glucocorticoid for which a prodrug is in clinical trials (Pfizer) inhibits mitochondrial potential similarly to Dex. The two SR compounds (Scripps Research) have pM affinity for GR and improve mitochondrial function. Not shown, SR1466 has better in vitro anti-inflammatory activity than PF802.



