BES2023 BES 2023 Section (29 abstracts)
A case of calcitriol-mediated hypercalcemia in a patient with solid tumor
Lomré M. , Sirtaine N. , Van Den Eynde K. , Kleynen P. , Martinez-Mena C. & Poppe K.G.
Case report: A 50-year-old man was referred by his family doctor to the emergency room in September 2020 for intermittent abdominal pain in the right upper quadrant for several months. He also mentioned weight loss and several episodes of fever with chills. The patient was an active smoker, consumed large amounts of alcohol, and had no known medical history, nor taking any medication. At physical examination, the patient appeared pale and cachectic, with tenderness on the right upper quadrant and slight defence, with no diminution of bowel sounds. Laboratory results showed microcytic anaemia (haemoglobin level of 7.6 g/dl) and severe - albumin adjusted - hypercalcemia (3.78 mmol/l). Protein electrophoresis showed no paraprotein (no spike/peak in the gamma zone) nor hypogammaglobulinemia. Biochemical workup (see Table 1) revealed suppressed PTH and PTHrP with elevated 1,25-dihydroxyvitamin. A contrast enhanced CT-scan of the abdomen revealed a heterogeneous mass of 12 cm of diameter in the liver with a possibility of kidney invasion accompanied by a dilatation of the intrahepatic bile ducts and a dilatation of the right kidney cavity. The same lesion was shown on the FDG PET-scan, without any other hypermetabolic lesion. On laparoscopic exploration, a tumoral mass invading the liver, the right kidney, the colon, the inferior vena cava and the duodenum with a walled retroperitoneum was seen. No resection was possible, and a biopsy revealed a sarcomatoid renal cell carcinoma. The patient was treated with hyperhydration and calcitonin. Due to remaining high calcium levels, zoledronic acid was administrated despite unfavourable dentist advice. Taking into account the possibility of a calcitriol-mediated hypercalcemia, 32 milligrams of methylprednisolone a day were initiated, with a certain degree of response. Denosumab was added given the new rise in calcium levels. Figure 1 shows the evolution of the calcium levels according to the treatment. The general condition of the patient continued to decline, and he sadly passed away. Staining for 1-alpha-hydroxylase was later performed on the biopsy, but remained negative.
| Parameters | Lab value |
| Total calcium | 3.78 (2.20-2.55 nmol/l) |
| Albumin | 4.1 (3.4-4.8 g/dl) |
| Phosphate | 0.62 (0.75-1.39 mmol/l) |
| Creatinine | 1.12 (0.70-1.20 mg/dl) |
| Intact parathyroid hormone (PTH) | < 10 (< 49 ng/l) |
| Parathyroid hormone-related peptide (PTHrP) | < 20 (< 20 pg/l) |
| Thyroid-stimulating hormone (TSH) | 1.99 (0.27-4.20 mU/l) |
| 25-hydroxyvitamin D | 19.9 (30-80 µg/l) |
| 1,25-dihydroxyvitamin D | 87.2 (29-83.6 ng/l) |
Discussion: Calcitriol-mediated hypercalcemia is a well-known entity in lymphomas and in granulomatous diseases (1,2). However, case reports of patients with solid tumors and calcitriol-mediated hypercalcemia remain scarce. Recently, Chukir and al. performed a retrospective study on 101 patients with solid tumors and hypercalcemia and showed that 45% of them had an elevated calcitriol, of which 76% also had an elevated PTHrP. This study also highlighted that those patients were rather poor responders to classical antiresorptive therapy, such as bisphosphonates or denosumab. (3) We observed similar findings in our patient. Two recent case reports of patients with calcitriol-mediated hypercalcemia showed an increased mRNAexpression of 1- h and vitamin-D receptors in cells from a gastrointestinal stromal tumor in one, and an expression of 1- h expression by the cells of a liposarcoma in another.(4,5) Our patient didn’t have increased PTHrP levels (lab testing was the same and consisted in an immunoradiometric assay on two PTHrP epitopes, without cross reactivity with PTH 1-84, PTH C-term and mid-term), but showed elevated calcitriol levels, suppressed PTH, and low phosphorus levels. No bone metastases were diagnosed, ruling out hypercalcemia due to osteolytic lesions. Staining for 1- h on the biopsy did not show any activity n the patient’s tumor. An argument in favour of this theory is the moderate response to high dosage of corticosteroids.
Conclusion: Physiopathology of hypercalcemia of malignancy is complex and might engage several mechanisms. Calcitriol-mediated hypercalcemia should be considered in case of solid tumors and complete biochemical workup remains crucial in order to better understand the underlying mechanisms and to predict poor responders to conventional antiresorptive therapy.Table 1. Biochemical workupFigure 1: Total calcium levels over timeAnti-CYP27B1 immunostaining (1-alpha-hydroxylase) on the primary tumor of the patient. Negative tumor cells ; note the positive staining in a normal macrophage, shown in the lower left image.
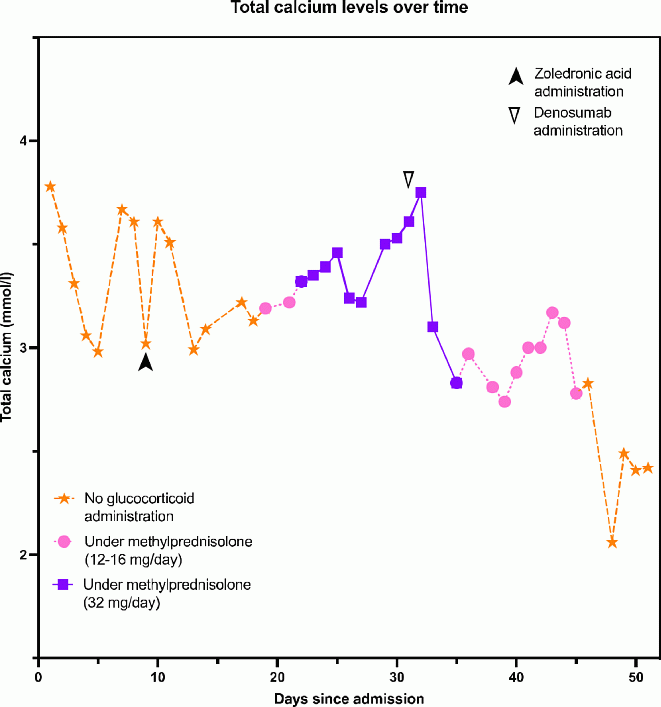
Figure 1. Total calcium levels over time

Bibliography: 1. Fuss M, Pepersack T, Gillet C, Karmali R, Corvilain J. Calcium and vitamin D metabolism in granulomatous diseases. Clin Rheumatol. 1992 Mar;11 (1):28-36.2. Seymour JF, Gagel RF. Calcitriol: The Major Humoral Mediator of Hypercalcemia in Hodgkin’s Disease and Non-Hodgkin’s Lymphomas. Blood. 1993 Sep 1;82c5):1383-94.3. Chukir T, Liu Y, Hoffman K, Bilezikian JP, Farooki A. Calcitriol Elevation Is Associated with a Higher Risk of Refractory Hypercalcemia of Malignancy in Solid Tumors. J Clin Endocrinol Metab. 2019 Dec 16;105(4):e1115 23.4. Herrera-Martínez Y, Contreras González MJ, Pedraza-Arévalo S, Guerrero Martínez MDC, Rodrigo Martínez Á, González Menchen A, et al. Calcitriol-Mediated Hypercalcemia, Somatostatin Receptors Expression and 25-Hydroxyvitamin D3- - Hydroxylase in GIST Tumors. Front Endocrinol (Lausanne). 2021;12:812385.5. Figueiredo A, Pereira T, Cabrera R, Simões-Pereira J, Leite V. Malignant Hypercalcemia Due to the Ectopic Production of Calcitriol by an Abdominal Liposarcoma. Cureus. 2023 Jan;15(1):e33446.

 }
}



